Accord Healthcare Inc. Issues Nationwide Voluntary Recall of Daptomycin for Injection 500 mg/vial and Daptomycin for Injection 350 mg/vial Lot # R2200232 Due to Product Mix-Up | FDA

Merck Issues Voluntary Nationwide Recall of CUBICIN® (daptomycin for injection) 500 mg, Lot 934778, Due to Presence of Particulate Matter Identified as Glass Particles | Business Wire
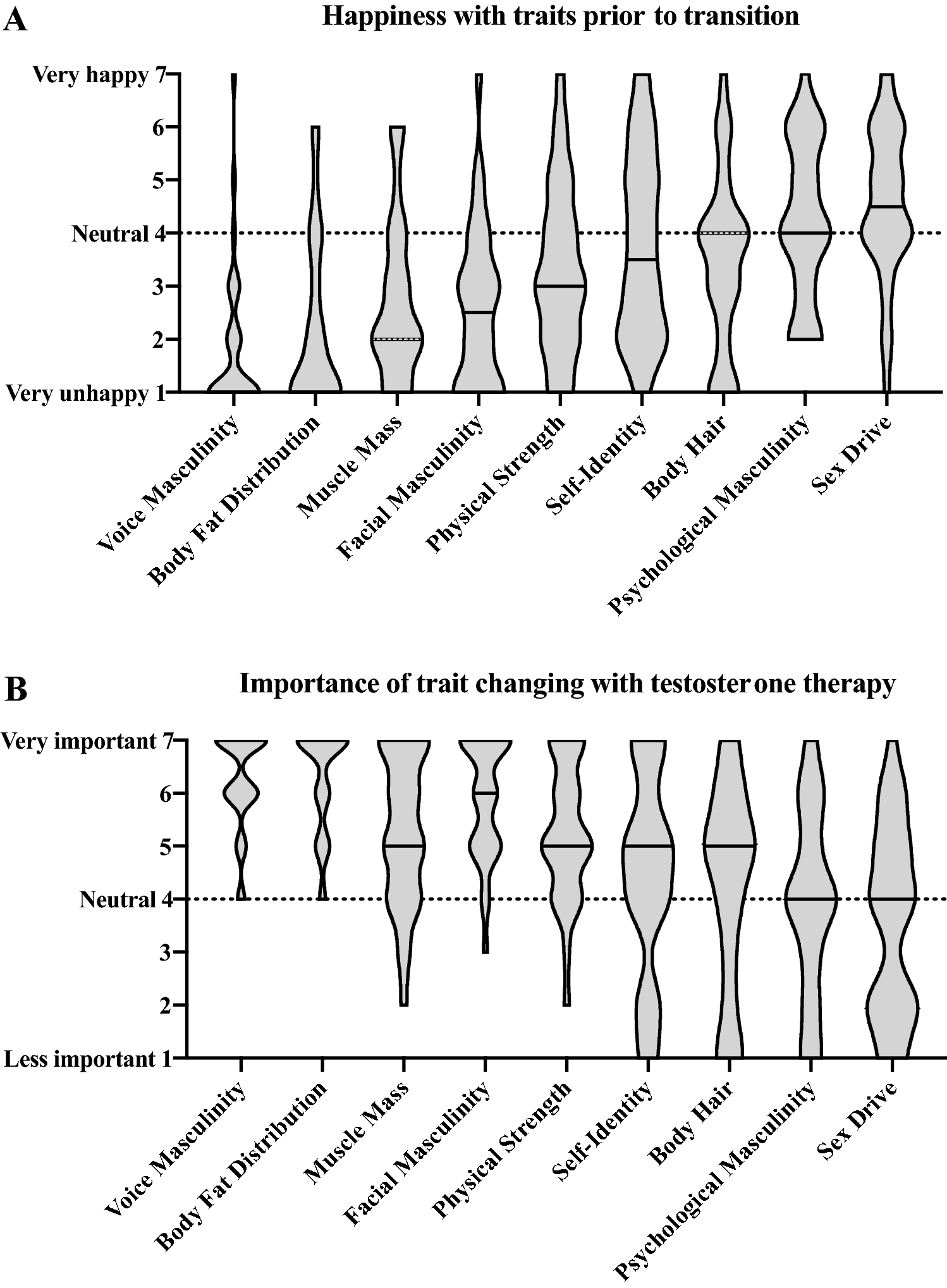

/cloudfront-us-east-1.images.arcpublishing.com/gray/J2OCFB7RRVBQDJRAB4KTAXNXNU.png)



/do0bihdskp9dy.cloudfront.net/08-20-2021/t_9ffac1ee93544b22808e8fc4d7a7264d_name_file_1280x720_2000_v3_1_.jpg)