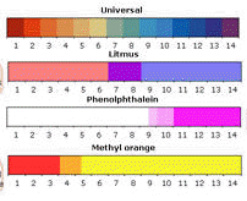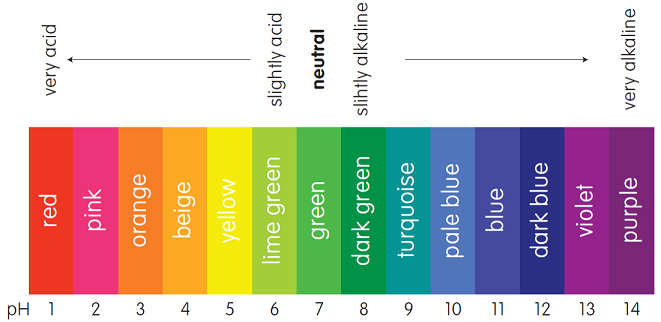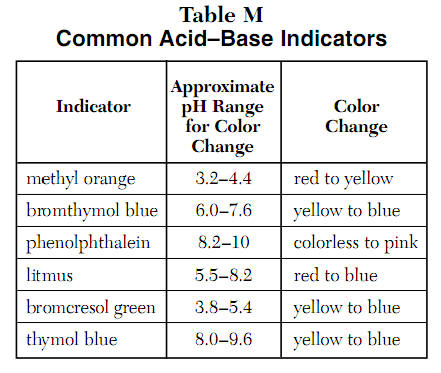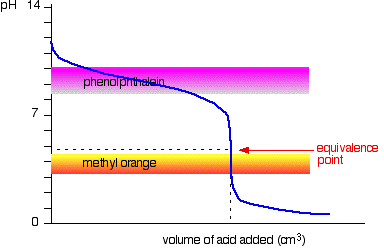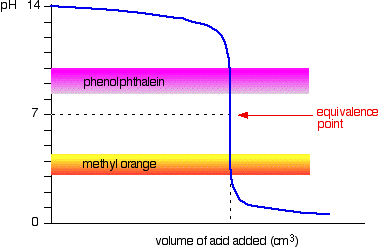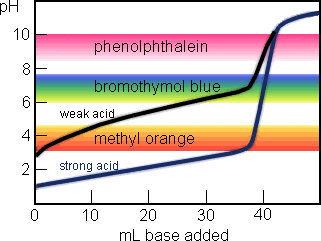
General Chemistry Online: FAQ: Acids and bases: How can strong and weak acids be distinguished using indicators?

Which one is the best indicator for acid and base titration between methyl orange and phenolphthalein? - Quora
A particular solution produced a yellow colour when tested with both cresol red, methyl orange and bromothymol blue. What does this imply about the pH of the solution? - Quora
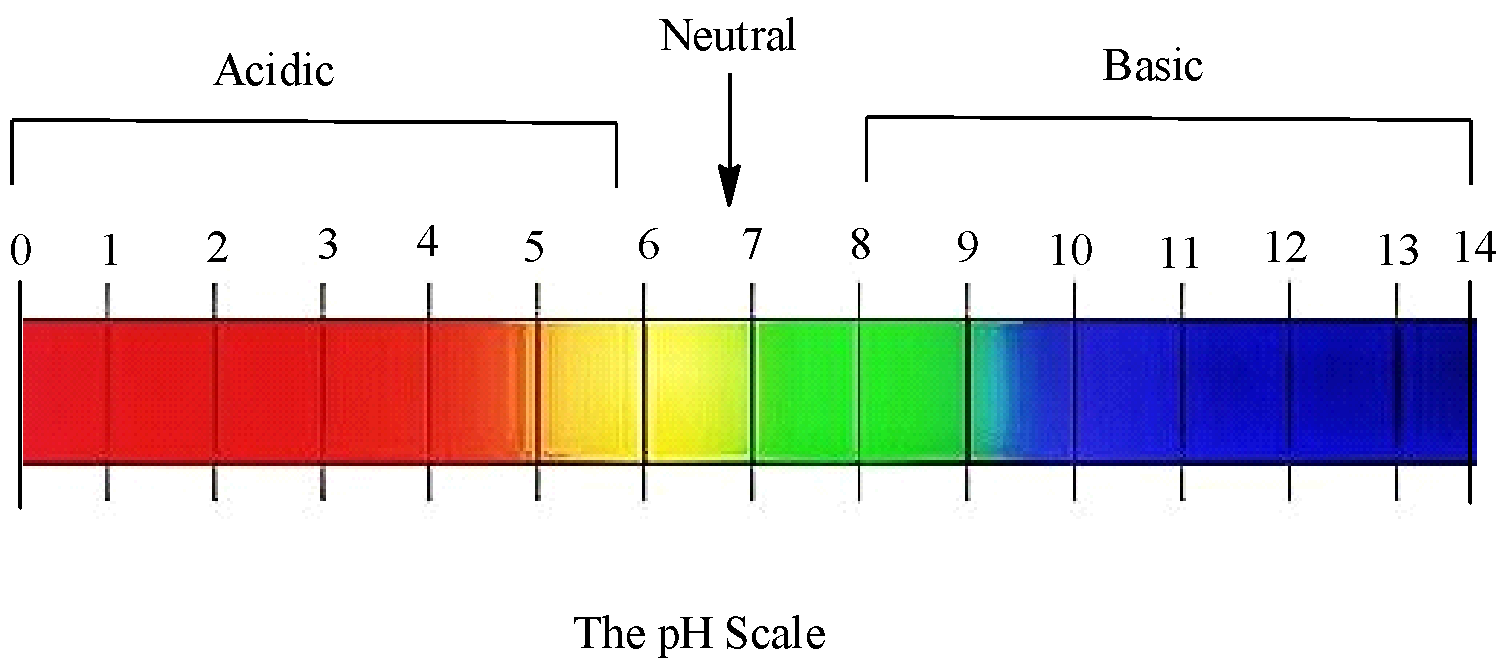
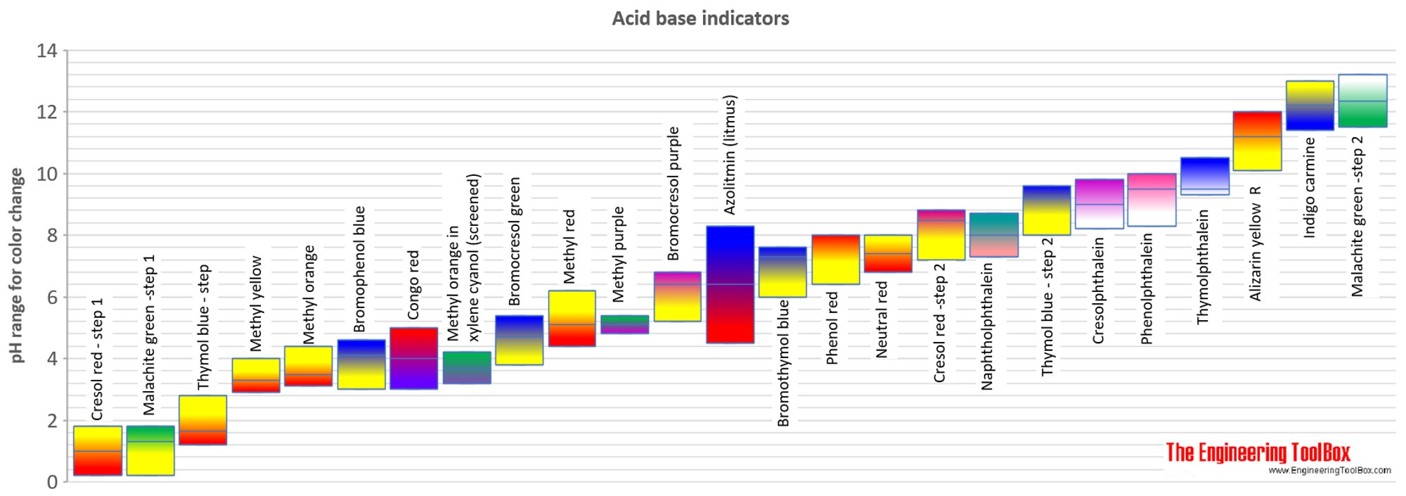


![Solved] At what pH value range does the methyl orange indicator chan Solved] At what pH value range does the methyl orange indicator chan](https://cdn.testbook.com/images/production/quesImages/quesImage8622.png)