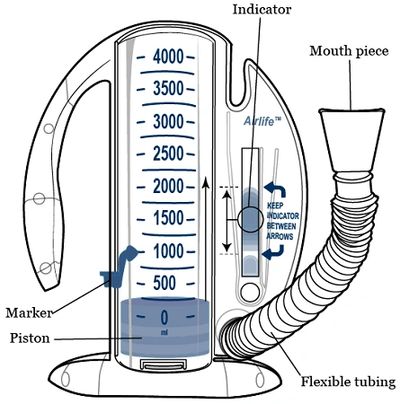
M MIR SmartOne Digital Spirometer - Peak Flow (PEF), FEV1 On Your Smartphone US FDA & CE approved with Free App & Reusable Mouthpiece Doctor's Most Recommended For Lung Health Check up :
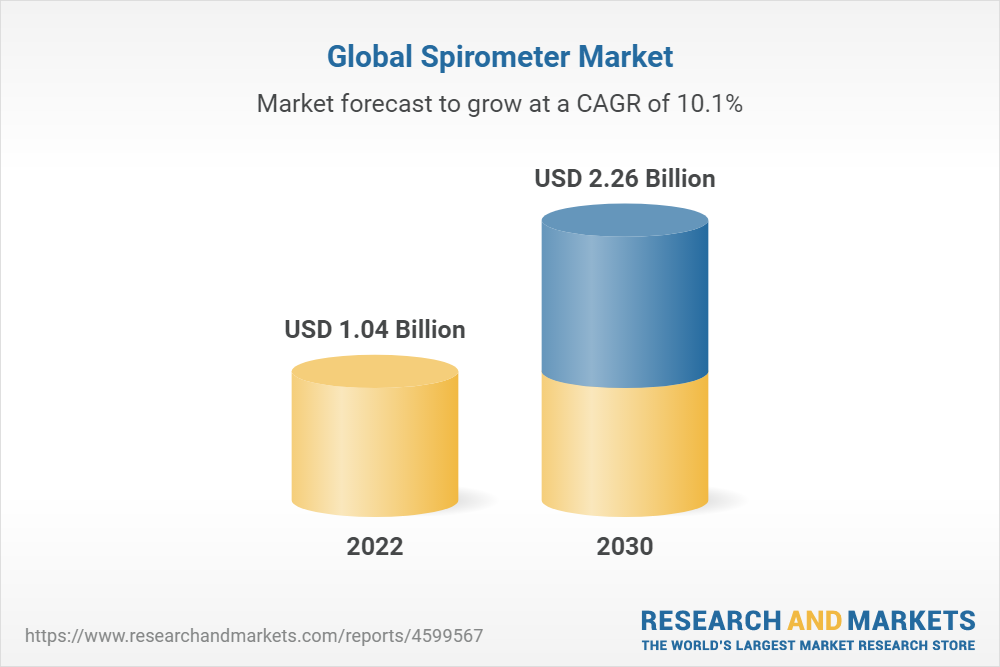
Spirometer Market Size, Share & Trends Analysis Report by Type (Hand Held, Table Top), by Technology (Volume Measurement, Flow Measurement), by Application (Asthma, COPD), by End Use, by Region, and Segment Forecasts,

Exploring the 175-year history of spirometry and the vital lessons it can teach us today | European Respiratory Society
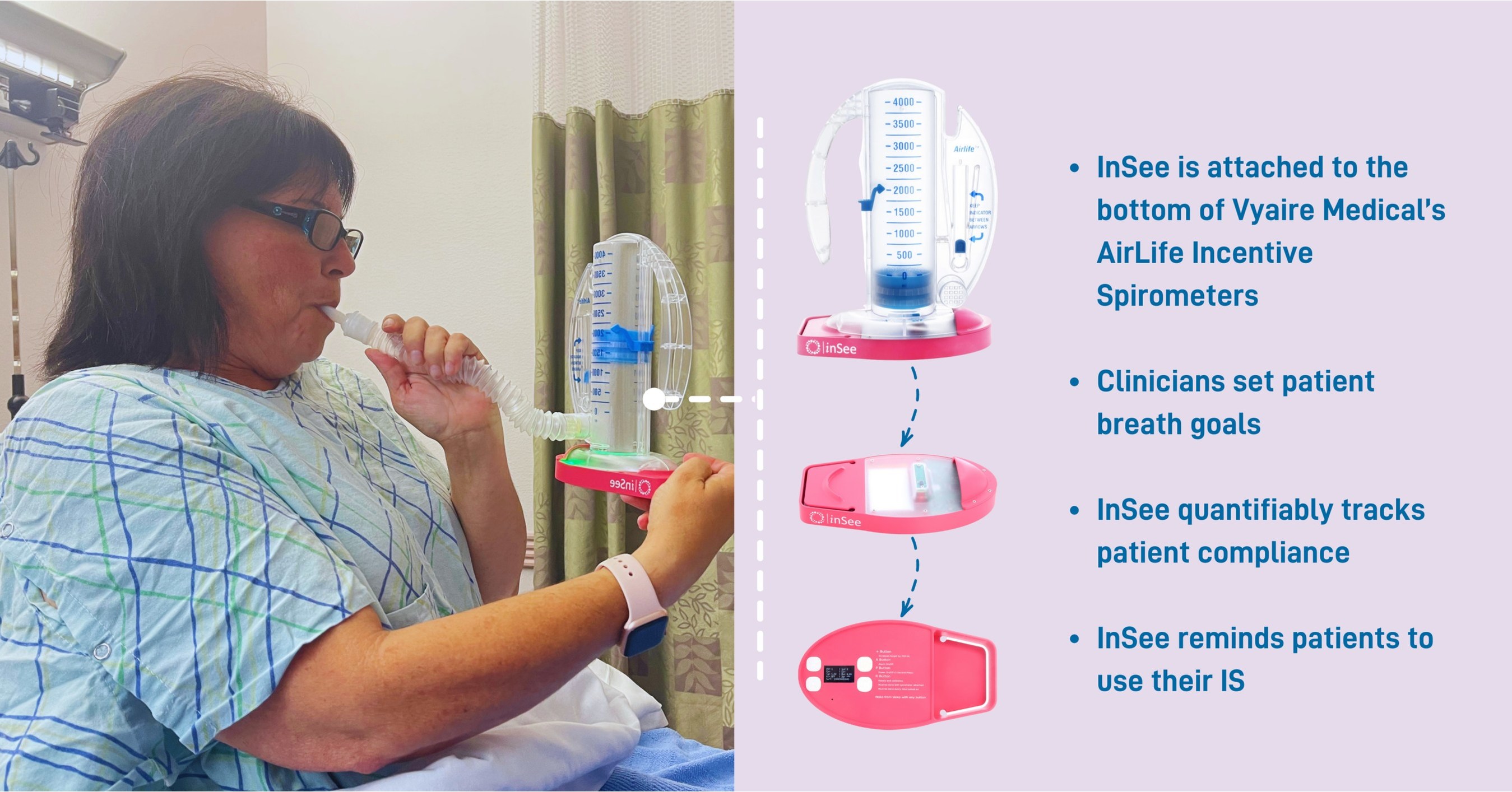
Tidal Medical Technologies receives FDA Emergency Use Authorization (EUA) for the use of InSee for tracking of incentive spirometers usage in treatment of respiratory conditions in COVID-19 patients.