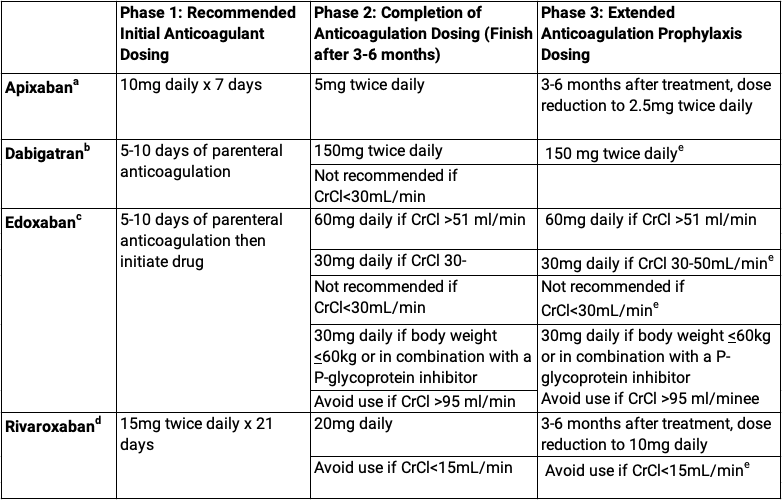
Apixaban (Eliquis®) – Drug-Drug-Interaction Considerations in COVID-19 Patients with Coagulation Abnormalities - Anesthesia Patient Safety Foundation

These highlights do not include all the information needed to use ELIQUIS safely and effectively. See full prescribing information for ELIQUIS. ELIQUIS® (apixaban) tablets, for oral useInitial U.S. Approval: 2012